A question people rarely ask
A plastic bottle can sit on a beach or in a landfill for years, and it still looks like a bottle. So it’s fair to wonder what it would mean for a microbe to “eat” it. This isn’t one single place or event. It’s been studied in labs and waste streams in a lot of regions, including Japan, the United States, and Europe. One well-known case came from a recycling-site sample in Japan reported in 2016, where researchers described a bacterium that could grow using PET-related material. The core idea is chemical, not magical: enzymes clip PET’s long chains into smaller molecules, and then the cell pulls those pieces into normal metabolism.
What PET is, at the bond level
PET is polyethylene terephthalate. It’s a polyester, which means its backbone contains ester bonds repeated over and over. Those ester links are the weak points bacteria can target, but “weak” is relative. PET is usually semi-crystalline. Some parts of the polymer are packed tightly (crystalline regions), and other parts are looser (amorphous regions). Enzymes can reach the looser regions much more easily, so the same bottle can be partly “available” and partly almost invisible to biology.
An overlooked detail is that PET products aren’t chemically identical. Processing and use change the surface. UV light, heat, and mechanical wear can add small cracks and oxidized groups. Even labels and additives can matter because they change how water sits on the plastic. Since these enzymes work at a wet surface, a tiny difference in surface hydration can change the reaction rate a lot.

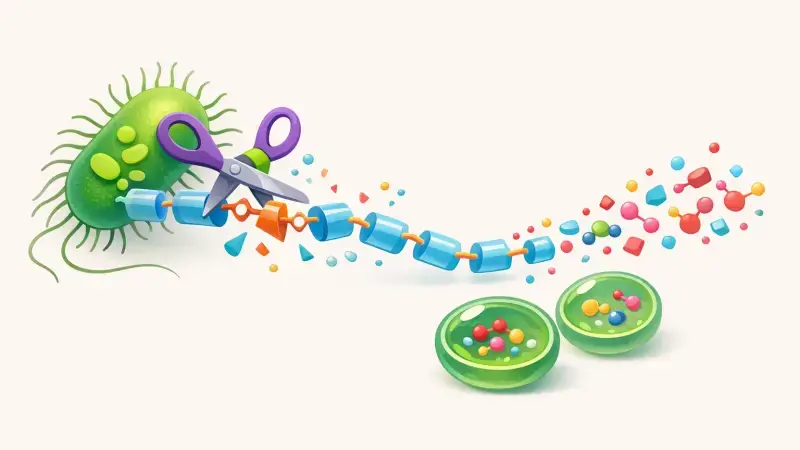
The first enzymatic cut: turning a solid into fragments
Bacteria that can attack PET tend to do it outside the cell. They secrete enzymes that stick to the polymer surface and hydrolyze ester bonds. The famous example from 2016 involved enzymes often described as PETase and MHETase. PETase makes the initial cuts and produces a mix of fragments. A key intermediate is MHET (mono(2-hydroxyethyl) terephthalate), along with smaller amounts of BHET and sometimes the final monomers directly.
Those cuts don’t happen “deep” inside the plastic. The enzyme can only reach what it can physically touch. That’s why temperature, agitation, and how finely the plastic is shredded can change results so much in experiments. The chemistry is the same, but the surface area and the mobility of polymer chains at the surface decide how many ester bonds are exposed long enough to be clipped.
Finishing the job: MHET to the monomers
The second step is turning those fragments into the simple building blocks a cell can actually use. MHETase (in that well-studied system) hydrolyzes MHET into terephthalic acid (TPA) and ethylene glycol (EG). This division of labor matters because MHET can build up and slow the first enzyme down. If MHET is removed efficiently, PETase can keep cutting without getting “stuck” behind its own products.
Once TPA and EG are free, the problem stops looking like “plastic” and starts looking like standard biochemistry. EG can be converted into central metabolic intermediates in several bacteria. TPA is aromatic and takes a more specialized set of steps, but some microbes have pathways that funnel it toward compounds that feed into the cell’s energy and carbon cycles. Different species use different enzymes here, and the exact genes vary, which is why lab results can differ even when the starting material is labeled simply as PET.
Why it’s slow in the real world
Most PET in daily life is designed to resist breakdown. Bottles and fibers have high molecular weight and significant crystallinity. The enzymes that can hydrolyze PET tend to work much better on amorphous PET, thin films, or pre-treated material. On an intact bottle, the accessible fraction is limited, and the surface can be coated with grime, biofilms, or minerals that block enzyme contact. Even when microbes are present, the plastic surface can be a poor place to live because it offers very little nitrogen, phosphorus, or trace metals.
A concrete situational example helps: a bottle in seawater may quickly grow a microbial film, but that doesn’t automatically mean the PET is being chemically cut. Many organisms in that film are eating dissolved organic matter in the water, not the polymer. Actual PET hydrolysis requires the right enzyme activity at the surface, enough water access to the ester bonds, and conditions where the fragments don’t just diffuse away faster than they’re produced.
If you liked this, you might also enjoy: