Underground life that doesn’t need oxygen
If you’ve ever seen rusty groundwater stain a sink or a culvert, you’ve seen iron chemistry leaking into daily life. Underground, some microbes run on that same chemistry, just without air. This isn’t one single place or event. It shows up in iron-rich aquifers in Bangladesh, in former mining regions like the Rio Tinto area in Spain, and deep below the seafloor at hydrothermal systems. The core trick is simple: instead of breathing oxygen, certain microbes pass electrons to metals and other minerals. That swap changes what dissolves, what precipitates, and what moves through rock.
What “breathing metals” actually means
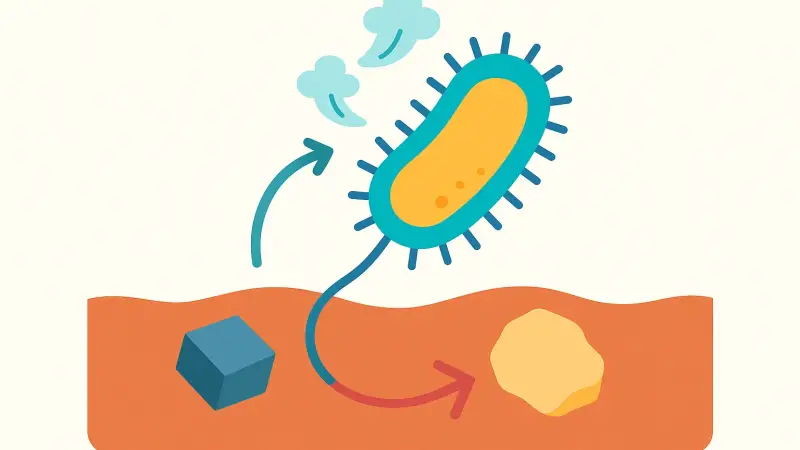
Breathing is really about moving electrons off food. Humans dump those electrons onto oxygen. Metal-breathing microbes dump them onto something else that can accept electrons, like iron(III) minerals (often written Fe(III)) or manganese(IV) oxides. When the microbe reduces Fe(III) to Fe(II), the mineral can change form or dissolve. That affects the water around it immediately. It can free up ions that were stuck to mineral surfaces, and it can change pH in small but important ways.
A specific overlooked detail is how physical contact matters. Some organisms, including well-studied genera like Shewanella and Geobacter, can move electrons outside the cell using outer-membrane proteins and sometimes conductive pili. Whether they must touch the mineral, or can use dissolved “electron shuttles” like humic substances, varies by microbe and by site. That tiny difference—touch versus shuttle—can decide how far their influence spreads through sediment pores.
How they rewrite subterranean chemistry
When iron and manganese minerals get reduced, groundwater chemistry can flip. One common change is that dissolved iron rises, which can later re-oxidize and form orange-brown precipitates when water meets air again. Another is that phosphate and trace metals that were adsorbed onto iron oxides can be released. That release is not a small side effect. It can change nutrient availability and shift which microbes can live there next.
Arsenic is a concrete, situational example people often hear about only in terms of “bad water,” not microbial respiration. In parts of Bangladesh and West Bengal, arsenic can be mobilized when iron(III) oxides that hold arsenic are reduced and dissolve. The exact mix of causes varies by aquifer, and it is not always clear how much is driven by microbes versus other geochemical inputs. But the mechanism—loss of the iron mineral “holding sites”—is central to why arsenic can move from sediment into water.
Minerals become habitats, not just background
Subsurface rocks aren’t smooth storage containers. They have grains, coatings, fractures, and tiny pores that create micro-environments. Metal-breathing microbes tend to cluster where electron acceptors are available, often right on mineral surfaces. That means the most active chemistry can happen in thin zones that a bulk water sample won’t represent well. Two centimeters of sediment can include oxygen-free pockets next to oxidized grain coatings, and the microbial community can track those boundaries closely.
Those boundaries also move. If organic matter arrives in pulses—after a flood, a seasonal recharge event, or a change in land use—the “redox ladder” in the sediment can shift. Iron and manganese reduction may intensify, then give way to sulfate reduction or methane production as different electron acceptors get used up. The chemistry isn’t marching toward a single endpoint. It keeps responding to supply lines: carbon coming in, minerals available, and water flow paths.
Why scientists care beyond the weirdness
Metal-respiring microbes sit at the center of practical questions because they control mobility. They can immobilize some contaminants by changing them into less soluble forms, and they can mobilize others by dissolving the minerals that were trapping them. Uranium is a well-known example in research: microbial reduction can convert U(VI), which is more soluble, to U(IV), which tends to precipitate. But local conditions can undo that later if oxidants return, so the stability of those minerals is site-dependent.
They also matter for how we read the deep past. Iron formations, mineral coatings, and isotope signatures can carry traces of microbial electron transfer. Interpreting those traces is tricky, because similar minerals can form abiotically. Still, when you find specific mineral textures alongside chemical gradients in sediments, it’s hard to ignore that microbes may have been “breathing” the rocks—quietly, persistently—while everything aboveground carried on.